How matrix recycling keeps the brain flexible
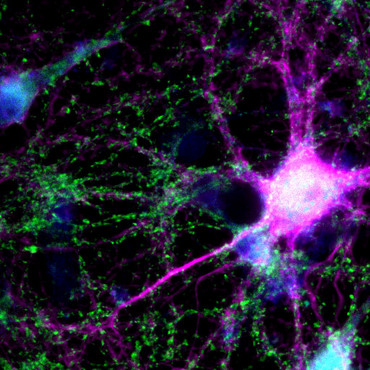
(mbexc/umg) The extracellular matrix (ECM) gives cell assemblies their structure and plays an important role in cell communication and control. In the adult brain, it forms lattices that sheath nerve cells and synapses. The frequent structural changes at synapses necessitate continuous remodeling of this lattice structure. Researchers at the Göttingen Cluster of Excellence "Multiscale Bioimaging: From Molecular Machines to Networks of Excitable Cells" (MBExC) now describe a new remodeling mechanism based on the recycling of individual components of the ECM that is closely linked to synaptic activity. This finding is relevant for clinical research, as a variety of brain diseases are associated with changes in the ECM. Recently, the results of the team around Prof. Dr. Silvio O. Rizzoli, Director of the Institute of Neuro- and Sensory Physiology at the University Medical Center Göttingen (UMG), speaker of the Board of the Center for Biostructural Imaging of Neurodegeneration (BIN) and member of the MBExC were published in the renowned journal Nature Communications.
Original publication: Extracellular matrix remodeling through endocytosis and resurfacing of Tenascin-R. Dankovich TM, Kaushik R, OlsthoornLHM, Cassinelli Petersen G, Giro PE, Kluever V, Agüi-GonzalezP, Grewe K, Bao G, Beuermann S, Abdul Hadi H, Doeren J, Klöppner S, Cooper BH, Dityatev A, Rizzoli SO (2021) Nature Communications,Nat Commun 12, 7129 (2021).
DOI: https://doi.org/10.1038/s41467-021-27462-7
Research results in detail
In the adult brain, the extracellular matrix forms a stable, mesh-like scaffold that envelops the nerve cells and synapses. The exeptional longevity of its components lends these lattices a unique durability. They are deemed to stabilize neural circuits, but thus also restrict their adaptability through remodeling (plasticity).
To ensure the function of the nervous system in the long term, the adult ECM retains the ability to be occasionally remodeled, in order to allow neural circuits to be altered throughout adulthood. In this process, the lattices are enzymatically cleaved near the synapses, followed by the secretion of newly-synthesized molecules that embed into the ECM, a fairly costly process for the cell metabolically.
Structural changes at synapses occur surprisingly frequently - on a time scale of minutes to hours. The authors' hypothesis: an additional, less energy-consuming remodeling mechanism must exist that relies on recycling instead of de novo sythesis of ECM molecules, which is closely linked to synaptic activity. This mechanism would lend the ECM the flexibility required for frequent synaptic changes.
To test their hypothesis of ECM recycling at synapses, the scientists focused on the glycoprotein tenascin-R (TNR), which is a known component of the extracellular matrix. And indeed, super-resolution fluorescence imaging combined with secondary ion mass spectrometry, revealed that a pool of mobile TNR molecules is enriched at synapses, and cycle in and out of the perisynaptic ECM via a surprisingly long route over a surprisingly long period of about three days. Even more, these molecules are transported in the body by neurons to the Golgi apparatus, where they presumably undergo remodeling by attachment of carbohydrates (glycosylation), and are then trafficked to synapses once again. Finally, a link between the extent of TNR recycling and synaptic activity could be established.